Written by Sarah Locknar
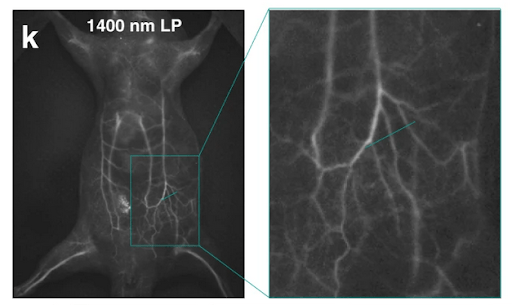
The whole-body fluorescence imaging of a mouse IV-injected with the NIR-II dye IDSe-IC2F encapsulated via PEGylation, excited with 793 nm laser light. Used under CC by 4 license without modification from (Feng, 2021) Figure 8k.
“Invisible” Imaging – Fluorescence Microscopy in the UV and NIR
As researchers push to extreme fluorescence multiplexing of 20+ labels per sample, they are reaching the limits of what can be accomplished in the visible wavelengths. One approach is cyclic-immunofluorescence microscopy, but another is to expand the useable wavelength range of fluorescent labels into ultraviolet (UV) and near-infrared (NIR) wavelengths. These wavelengths provide unique challenges to the experimental setup as discussed below.
UV fluorescence microscopy
UV wavelengths span 1 – 400 nm. For biological applications they are grouped into UVA, UVB, and UVC (Figure 1) largely based on what sort of damage they cause in tissues (eyes and skin). Shorter UV wavelengths are termed extreme UV (EUV) and are used in the semiconductor industry. UVA fluorescence microscopy is relatively common, including imaging of the intrinsic chromophore NADH (Figure 1), Calcium indicator Indo-1, and the nuclear dye DAPI. Other options can be found with FluoroFinder’s Dye Directory and Spectral Viewer.
Double-stranded DNA, RNA, and the amino acids tryptophan, phenylalanine, and tyrosine absorb UVB/C radiation (Figure 1), which can induce photodamage directly or indirectly by free-radical generation. Few biological researchers work in UVC wavelengths because of this damage potential and rapid photobleaching. Nevertheless, UV-induced photochemistry can be advantageous for uncaging procedures where focused light releases molecules of interest quickly in a known location.
Mie light scattering is also a problem at UV wavelengths because the size of subcellular components (organelles, vesicles, etc.) are similar to the wavelength of light. This is the same phenomenon that causes clouds to appear white. Scattering limits the penetration of excitation light into tissues, so single cells, monolayers, or thin tissue sections must be used.
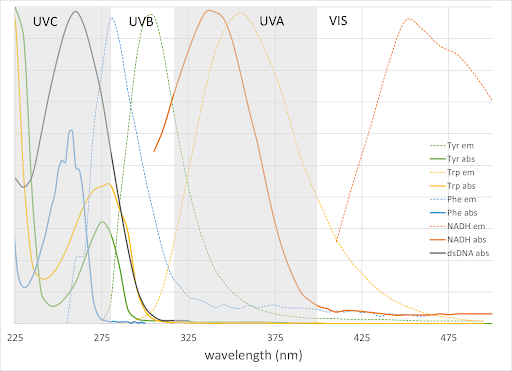
Figure 1. Absorption and emission of amino acids, NADH, and double-stranded DNA in the UV range. Visible, UVA = 315-400nm, UVB = 280-315nm, UVC 100-280 nm regions are also indicated.
Hardware for UV fluorescence microscopy
Light sources– Traditional light sources for UV imaging have been Deuterium and Mercury lamps, which emit wavelengths down to about 200 nm. The relatively short lifespan of Deuterium lamps, disposal issues with Mercury, and rapid development of LEDs (especially for UV sterilization during COVID) have largely replaced these lamps. There are also lasers available at 261, 349, 351, and 375 nm for confocal applications. Some microscopy systems, especially those designed for wide-field live-cell imaging, incorporate a UV-blocking filter in the optical path that must be removed for UV imaging experiments.
Detectors – Standard visible sensors are intrinsically UV sensitive, but their response is reduced because many glasses absorb light at wavelengths less than 350 nm (Figure 2). Replacing glass with fused silica or other materials such as fluorite allows transmission and detection into UVC. Figure 2 includes normalized quantum efficiency curves for representative UV-sensitized PMT and CMOS cameras.
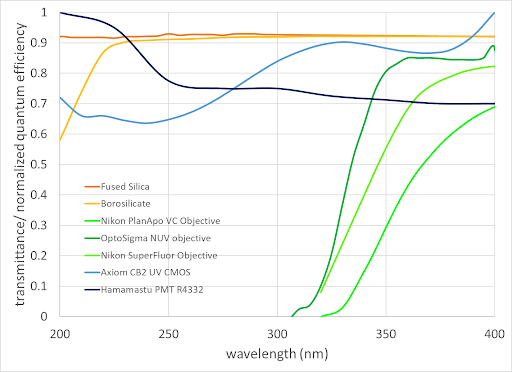
Figure 2. Transmittance of some optical materials and objective lenses and normalized QE of UV-enhanced PMT and CMOS detectors
Objective lenses – To be used in fluorescence microscopy, an objective lens must transmit both the excitation and emission wavelengths efficiently. As mentioned above, many glasses stop transmitting at about 350 nm. Researchers can increase the excitation light intensity to compensate for low throughput, but this can cause heating and degradation of the objective lens over time.
Light transmission is only part of the story for an objective lens used in multiplexed fluorescence microscopy. To ensure image integrity, chromatic aberrations (different colors focusing at different focal planes) between the UV and visible signals should be minimized. Standard apochromat lenses are only corrected in the visible range (usually ~ 435-660 nm). Apochromats with 4-color correction are available at 405 nm (Nikon CFI plan apo VC 60XC WI, Figure 2) and at 355nm from (OptoSigma NUV objective, Figure 2). The semiconductor metrology field is also actively working on new UV and EUV lens designs, including reflective lenses that do not exhibit chromatic aberrations. Note that using objectives from a third party (instead of the microscope manufacturer) may result in unexpected differences in magnification, field-of-view, and/or parfocality.
The lack of suitable objective lenses is a major impediment to UVB/UVC fluorescence microscopy. Researchers can sometimes get around this by using pulsed multiphoton excitation techniques as described in the last section.
Near-infrared (NIR) fluorescence microscopy
NIR wavelengths include those from about 700 – 3000 nm. NIR imaging is useful because there is less autofluorescence, and it can deeply penetrate tissue without being significantly scattered. One issue in NIR imaging is the absorption of water at several wavelengths (Figure 3), which causes sample heating and reduced light penetration at those wavelengths. The regions between the major water absorption bands are called “NIR windows” as indicated in Figure 3. Note that the wavelength cutoffs of these regions vary somewhat in the literature, especially for NIR-II, which, as illustrated in Figure 3, spans the 1000-1700 nm range. (Lane, 2018) Some scientists (usually from the military and satellite imaging communities) also lump the wavelength ranges into NIR (700-1000 nm) and SWIR (short-wave IR, 1000-3000 nm).
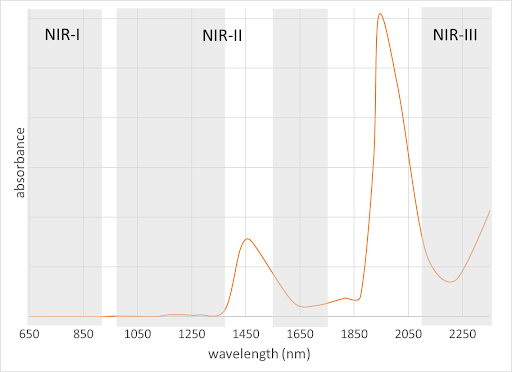
Figure 3. Water absorption peaks and NIR windows. NIR-II is broken into two subregions around the 1450 nm water absorption band.
NIR-I absorbance is widely used for clinical applications such as pulse-oximetry monitoring and vein-finding. (Miyake, 2006) Both are based on hemoglobin and/or oxy-hemoglobin absorption at 660 nm and 940 nmm, respectively. Oxygenation levels are calculated using ratios of these two absorption bands. NIR-I fluorescence is also common in whole-animal imaging systems used in biological and pharmaceutical research (cover image). Many commercially available NIR-I fluorescent dyes can be found using FluoroFinder’s Spectra Viewer. Most fluorescence and confocal systems on the market are suitable for NIR-I excitation and detection.
Options for NIR-II dyes are more limited (Figure 4). Designing small-molecule dyes at longer wavelengths is problematic, mostly due to twisting of the molecular structure. However, some options are available, including from Sigma-Aldrich and CD Bioparticles. Aggregation-induced emission (Liu, 2022) is one way to reduce twisting and increase fluorescence signal. Another approach uses nanoparticles (gold, rare earth, or carbon-based) and quantum dots. (Dunn, 2023) Lead sulfide quantum dots are available with peak emission spectra up to 1800 nm (CD Bioparticles, Millipore Sigma). NIR-II dye development is an active area of research, with new solutions becoming available every year. New molecular design strategies are even enabling some probes with emission tails into the NIR-III, which has very few options. (Meador, 2024)
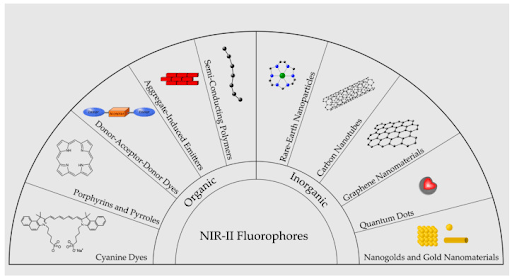
Figure 4. Illustration of major categories of NIR-II emitting fluorophores. Used under CC by 4 license without modification from (Dunn, 2023) Figure 2.
Hardware for NIR fluorescence microscopy
Light sources – The quartz Tungsten halogen (QTH) lamps (filament-based) provide stable, reliable blackbody radiation through NIR-III. These traditional light sources are gradually being replaced with LEDs that are available up to about 1600 nm (NIR-II). Diode lasers at 730 (Zeiss LSM 990) and 785nm are options on some commercially available confocal microscopy systems (Nikon AX/AXR with NSPARC, Evident FV3000). There are many other laser options available, including rare-earth fiber lasers in tunable configurations that span the NIR-I-III range.
Detectors – The NIR-I region is covered by standard Si-based cameras and PMTs, which has led to rapid adoption. The NIR-II signal is best detected with InGaAs-based APDs and cameras (Hamamatsu, Raptor). NIR-III detectors are typically based on InAs or PbS (Figure 5). HgCdTe can also be used for NIR-III and longer wavelength detection (> 5000 nm). Many NIR-II and NIR-III detectors use thermoelectric or liquid nitrogen cooling to reduce electronic noise in the systems.
Objective lenses –Super-achromat objective lenses are available from most major microscope manufacturers with chromatic aberration correction through the VIS and NIR-I regions for multiplexed imaging. There are also apochromats with correction in the NIR-II region available. Again, note that using third party objectives may lead to unexpected consequences. NIR-III objectives are not commercially available at this point in time.
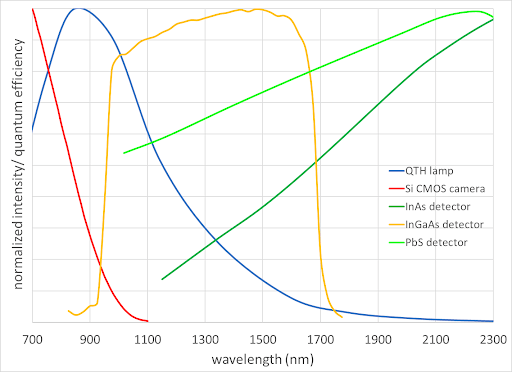
Figure 5. NIR emission of QTH lamps and normalized detector quantum efficiency curves
Multiphoton imaging – NIR excitation, UV emission
Multiphoton microscopy uses a high-powered femtosecond pulsed NIR laser to deliver multiple photons simultaneously to fluorophores at the focal point. Available lasers include traditional tunable Titanium: Sapphire lasers and fiber lasers throughout the NIR regions. Excitation occurs at roughly ½ or 1/3 the laser wavelength for two-photon or three-photon excitation respectively. Because the required power density only occurs in a very small volume, all emitted light is collected. This means that chromatic aberration correction of the objective is not important unless more than one excitation wavelength is used (super-apochromatic objective lenses are preferred). Nevertheless, high transmission is required from UV-NIR to detect UVA/UVB emitting molecules. (Zipfel, 2003)
Summary
Imaging at the extremes is an active area of growth in fluorescence microscopy. UVA and NIR-I regions are already well-established options, while research continues outside of these regions. UVB and UVC are limited by scattering and photodamage concerns, while in the NIR, new dye, laser, and optical materials development continues to push the envelope into the NIR-III, not only for the research scientist, but for surgical applications as well.
References
Dunn, H. H. (2023). doi:10.3390/bioengineering10080954
Feng, T. W. (2021). doi:10.1038/s41377-021-00628-0
Lane, X. N. (2018). doi:10.1016/j.cbpa.2018.03.015
Liu, L. Z. (2022). doi:10.1016/j.matt.2022.10.005
Meador, L. L. (2024). doi:10.1038/s41557-024-01464-6
Miyake, Z. D. (2006). doi:10.1111/j.1524-4725.2006.32226.x.
Zipfel, W. C. (2003). doi:10.1073/pnas.0832308100