When designing a flow cytometry experiment, it is important to account for the relative brightness of each fluorescent label on your specific instrument. Ideally, brighter fluorophores should be assigned to weakly expressed markers, while dimmer fluorophores should be assigned to highly expressed markers. However, a fluorophore’s brightness can be influenced by various factors, including signal intensity, background autofluorescence, non-specific staining, specificity, and more. Subsequently, researchers require a method to normalize these factors and determine the relative brightness of their fluorophores.
The Stain Index
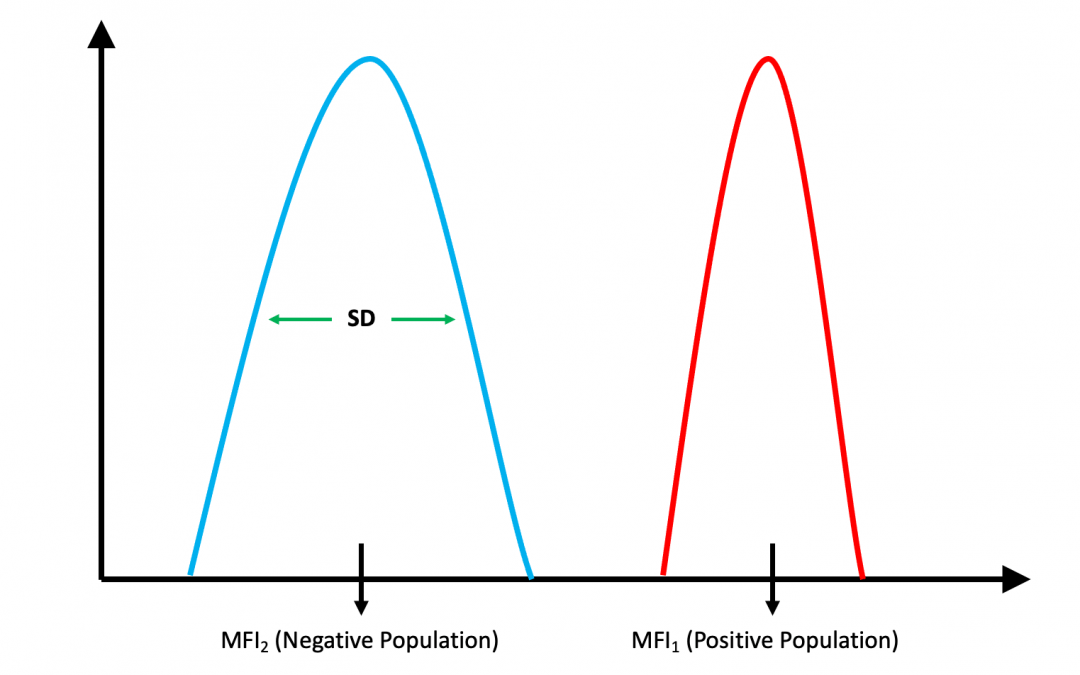
The stain index measures the relative brightness of various fluorophores on a given cytometer. This value can be used to compare fluorophores’ brightness to assign them appropriately to their flow cytometry panel.
To determine the stain index, a panel of different fluorophores conjugated to the same clone is tested side by side on the same cytometer. This experiment can be conducted using a widely expressed marker with commonly available antibodies that come in various fluorophores.

The stain index is defined as the difference between the mean fluorescence intensity of the positive and negative populations, divided by two times the standard deviation of the negative population.
Brightness Rating vs Stain Index
 Fluorophore suppliers typically provide a brightness rating from one to five to serve as a guide to compare relative brightness. For example, FITC has a brightness rating of 2, DyLight™ 488 has a brightness rating of 3, and VioBright FITC has a brightness rating of 5. FluoroFinder’s Panel Builder displays these ratings during the reagent selection process to help researchers quickly assess the relative brightness of their fluorophores. During marker selection, users can also adjust the antigen expression level of their markers to filter available fluorophores by brightness.
Fluorophore suppliers typically provide a brightness rating from one to five to serve as a guide to compare relative brightness. For example, FITC has a brightness rating of 2, DyLight™ 488 has a brightness rating of 3, and VioBright FITC has a brightness rating of 5. FluoroFinder’s Panel Builder displays these ratings during the reagent selection process to help researchers quickly assess the relative brightness of their fluorophores. During marker selection, users can also adjust the antigen expression level of their markers to filter available fluorophores by brightness.
 However, it is important to note that each supplier may use their own criteria to determine their fluorophores’ values for relative brightness. Therefore, these ratings should be considered as a general reference rather than an absolute measure. To account for the wide selection of fluorophores and variations between cytometers, we recommended that researchers calculate their own stain index. This is particularly crucial for sensitive experiments and when using a new fluorophore.
However, it is important to note that each supplier may use their own criteria to determine their fluorophores’ values for relative brightness. Therefore, these ratings should be considered as a general reference rather than an absolute measure. To account for the wide selection of fluorophores and variations between cytometers, we recommended that researchers calculate their own stain index. This is particularly crucial for sensitive experiments and when using a new fluorophore.
Stain Index and Titration
Stain index values are also useful for evaluating titrations to find optimal antibody concentrations. Plotting the titration data using stain index vs concentration can be used to better visualize the titration calculation.
The stain index is critical for researchers to take into account when designing their experiments. Make sure that you are considering the staining index and avoiding these common mistakes researchers make when designing flow cytometry antibody panels. Consider consulting your flow core manager, or delegated core contact if you have questions about the stain index for your specific cytometer.