Antibody conjugate panels must be designed with care to ensure accurate results
Immunophenotyping is a flow cytometry-based technique which uses fluorophore-labeled antibodies for identifying different cell types within a heterogeneous population. While some cell types can be identified using just a single marker, others require multiple markers to be detected simultaneously. As the number of markers increases, optimizing a flow cytometry immunophenotyping assay becomes more challenging. However, by following best practices and using online tools such as FluoroFinder’s Panel Builder, researchers can achieve reliable results.
What does a typical immunophenotyping assay involve?
A typical immunophenotyping assay begins with collecting sample material – usually blood or excised tissue – and preparing a single-cell suspension. The cells are then stained with antibodies targeting surface antigens, or are fixed and permeabilized before staining for intracellular targets. The immunostaining process involves a series of critical steps: blocking to prevent non-specific antibody binding, incubation with primary antibodies, then washing and incubation with secondary antibodies if these will be used for detection. After additional washing to remove any unbound secondary antibody reagents and resuspension of the immunostained cells in a suitable buffer, the sample is introduced into the flow cytometer for identification of different cell types based on light scatter and fluorescence.
Tips for optimization
Like any immunoassay, immunophenotyping requires careful planning and optimization, and researchers must consider everything from the sample type being investigated to necessary controls. Here are our top five tips for immunophenotyping success:
Understand your experimental system
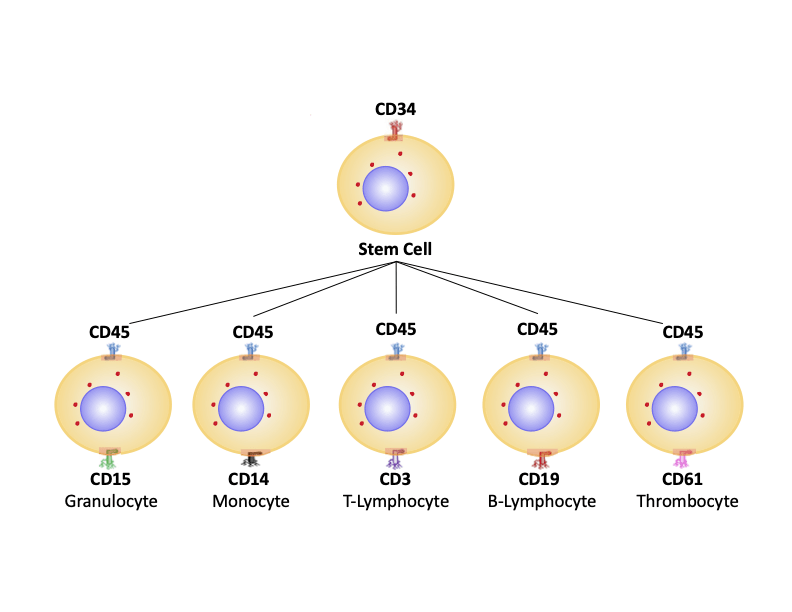
Learning as much as possible about the biology of the system you plan to study is an important first step in any immunophenotyping experiment. This should include establishing which markers are required for definitive identification of different cell types, understanding the expressed markers’ locations on the cell, and determining whether treatment is needed to stimulate marker expression. Sites such as UniProt, PAXdb, and The Human Protein Atlas are useful resources for answering these questions, as are antibody datasheets and literature searches. If a combination of extracellular and intracellular markers is required to identify a particular cell type, it is often recommended to use a sequential staining protocol, whereby extracellular markers are detected first to avoid antibody epitopes being damaged during fixation and permeabilization.
Ensure you have a single-cell suspension
For correct interpretation of flow cytometry data, it is essential that sample material is in the form of a single-cell suspension. If two or more cells simultaneously cross the flow cytometer’s interrogation point, they will mistakenly be identified as a cell type which expresses all of the detected markers. Best practices for preparing a single-cell suspension include adding EDTA, which chelate metal ions involved in cell adhesion, and DNase, which digest free DNA. It is also recommended to use a resuspension buffer that is free of Ca2+/Mg2+ and pass samples through a cell strainer before loading them into the flow cytometer.
Take care with panel design
Panel design is the process of assigning antibodies and fluorophores to markers that will be used for cellular identification. Antibodies should be purchased from a trusted vendor and validated for flow cytometry. It is also suggested that researchers use the manufacturer’s recommended staining protocol (provided on the antibody datasheet) as a starting point for optimization in their own model system. Fluorophores should be spectrally distinct from one another and feature excitation and emission maxima that are compatible with the flow cytometer’s configurations. To ensure that both scarce and highly expressed markers are detected, bright fluorophores should be paired with low density antigens, and dimmer fluorophores with more abundant targets.
Include relevant experimental controls
Flow cytometry experiments require several controls in addition to the usual positive and negative samples incorporated into a well-designed immunoassay. These include unstained controls (samples which have not been incubated with antibody reagents) for assessing autofluorescence and light scatter; fluorescence-minus-one controls (samples that have been stained with all but one of the fluorophores in the immunophenotyping panel) for evaluating fluorescence spread and setting gates, and compensation controls (samples stained with just a single fluorophore) for addressing spectral overlap. Researchers may also use isotype controls for investigating non-specific antibody binding at the cell surface, and Fc blocking controls for confirming that an Fc blocking step (the use of specialized reagents to prevent antibody binding to Fc receptors found on monocytes, macrophages, dendritic cells and B cells) has been effective.
Be consistent
Once panel design and protocol optimization are complete, it is important to be consistent with how samples are prepared and handled, and how flow cytometry data is analyzed. Not only will this safeguard experimental reproducibility, but it will also help with transferring the assay to other researchers. Protocol details should be clearly documented, and the panel recorded for future reference, and it is also recommended that reagent lot numbers be monitored over time to enable rapid identification of any potential sources of variation.
Resources for flow cytometry immunophenotyping
No matter how complex your flow cytometry immunophenotyping assay, FluoroFinder is here to make your life easier! Use our Antibody Search function to find flow cytometry-validated antibodies for your targets of interest, then turn to our Spectra Viewer to compare the properties of over 1,000 fluorophores in a single resource. For panel design, our Panel Builder lets you view the fluorophore and antibody offerings from all major suppliers to help you ensure the validity and reproducibility of your experiments.
Sign up for our eNewsletter to receive further updates about flow cytometry immunophenotyping assays and other fluorescence-based techniques.