Written by Paul E. Mead, BSc (Hons), PhD, SCYM (ASCP)
Clinical Flow Cytometry: Diagnosis and Applications
From Bethesda consensus to high-dimensional cytometry and the next era of clinical diagnostics
What Is Clinical Flow Cytometry?
Clinical flow cytometry is a laboratory technique used to analyze the physical and chemical characteristics of cells, most commonly used in diagnosing hematologic malignancies, monitoring immune disorders, and assessing treatment response.
Many of the most consequential decisions in medicine begin with a measurement. A physician makes a diagnosis, adjusts therapy, predicts prognosis, or reassures a patient based on laboratory results that must accurately reflect biological reality. In hematologic disease, clinical flow cytometry plays a central role in generating these measurements. Although often associated with lasers, fluorochromes, and increasingly sophisticated instrumentation, its purpose extends beyond the technology itself. Clinical cytometry transforms complex cellular patterns into information that clinicians can use to guide patient care.
Why Measurement Matters in Clinical Diagnostics
A measurement is only as valuable as it is reliable. In clinical medicine, consistency, reproducibility, and interpretability are essential. Early advances in flow cytometry provided powerful tools for identifying abnormal cell populations. However, without standardization, even accurate results could lack comparability across laboratories. As a result, inconsistency can undermine confidence and limit clinical utility, particularly in settings where treatment decisions depend on subtle distinctions.
From Innovation to Inconsistency
By the early 1990s, immunophenotyping had already become central to diagnosing diseases such as acute lymphoblastic leukemia and other hematologic malignancies. Laboratories could characterize leukemic cells with high accuracy, fundamentally changing diagnostic workflows. At the same time, variability persisted across institutions. Different laboratories used distinct antibody panels, applied different gating strategies, and reported findings in ways that were not always aligned.
Consequently, even high-quality results were not always directly comparable. In clinical diagnostics, where consistency underpins trust, this lack of alignment represented a significant limitation.
The Bethesda Consensus: Standardizing the Field
A major turning point came with the Bethesda Clinical Flow Cytometry Conferences in 1995 and 2006. Importantly, these meetings shifted the focus from expanding technical capability to standardizing how cytometry is applied in clinical practice.
Rather than emphasizing increasingly complex panels or new markers, the field aligned around consistency. Consensus efforts established structured reagent frameworks, clarified approaches to data interpretation, and promoted harmonized reporting practices. Ultimately, the goal was to ensure that measurements carried the same meaning regardless of where they were generated.
The outcomes of these meetings shaped many of the foundations of modern clinical cytometry. Consensus reagent frameworks were established for evaluating major cellular lineages, including B cells, T and NK cells, and myelomonocytic populations. These recommendations provided structured diagnostic starting points while still allowing laboratories flexibility for expanded analyses.
Equally important were improvements in reporting practices. Standardized terminology for antigen expression and greater emphasis on fluorescence intensity distributions enhanced communication between laboratories and clinicians. These changes reduced ambiguity and improved the comparability of results across institutions.
From Laboratory Technique to Clinical Infrastructure
As standardization took hold, flow cytometry evolved from a specialized laboratory technique into a core component of clinical diagnostic infrastructure. Laboratories operating within a shared measurement framework could generate results that were not only accurate but also comparable across institutions.
Expanding Clinical Impact
As a result, this shift enabled more reliable multi-center clinical trials and strengthened risk stratification models used in patient care. It also enhanced the clinical value of assays designed to detect minimal residual disease, where small differences in measurement can influence critical treatment decisions.
While improvements in outcomes, such as increased survival rates in pediatric leukemia reflect advances across therapies and care systems, flow cytometry has played an essential supporting role by providing clinicians with dependable measurements.
Regulation, Accreditation, and Quality Systems
The principles established through consensus efforts influenced how clinical laboratories operate under regulatory frameworks. Flow cytometry is classified as high-complexity testing under CLIA requirements.
In practice, laboratories must meet rigorous standards for validation, quality control, and personnel competency. Guidance from the Clinical and Laboratory Standards Institute helps define best practices for assay validation and ensures that measurements remain consistent, reproducible, and clinically reliable.
In addition, accreditation bodies such as the College of American Pathologists reinforce these expectations through structured inspection programs that emphasize assay performance, reagent validation, and instrument consistency.
Lessons from HIV: The Importance of Reliable Measurement
The importance of reliable measurement in cytometry became especially clear during the HIV/AIDS epidemic. Clinicians needed dependable methods to monitor immune function in patients whose disease progression could change rapidly. CD4+ T-cell counts emerged as a critical biomarker, guiding both prognosis and treatment decisions.
Importantly, meeting this need required more than technological capability. It demanded standardized protocols, shared reference materials, and consistent analytical approaches. As such, the experience reinforced a principle that continues to guide clinical diagnostics today: innovation alone is insufficient without standardization.
Technological Expansion and the Rise of High-Dimensional Cytometry
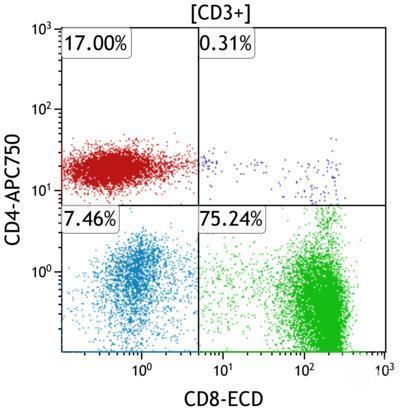
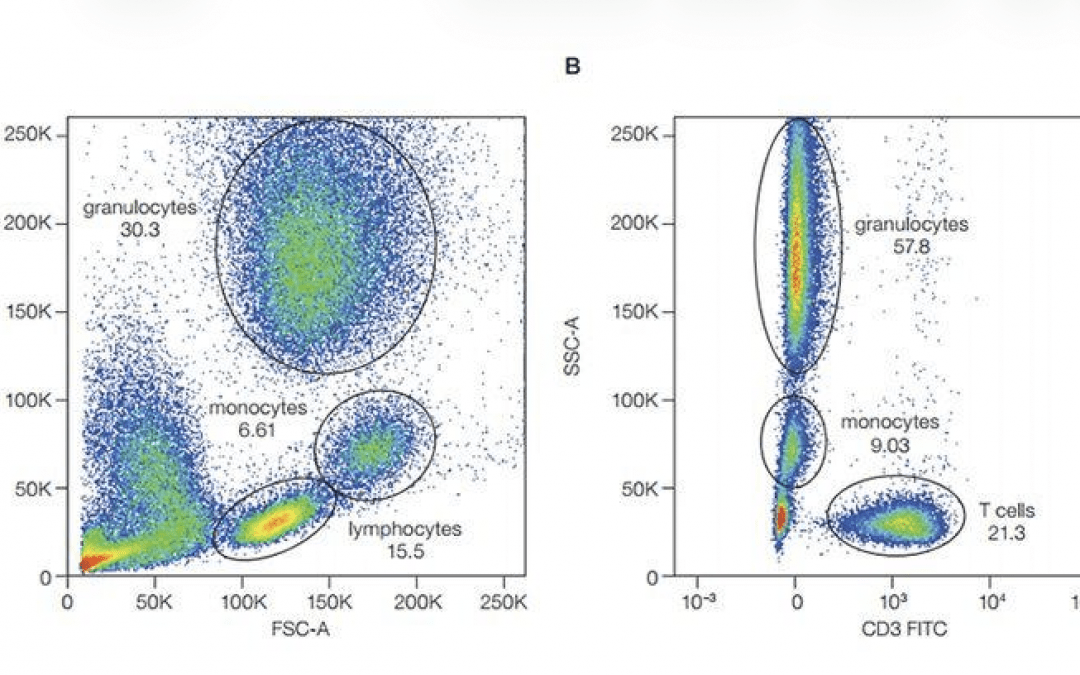
Figure: Flow cytometry dot plot used to identify and gate distinct cellular populations in clinical diagnostics.
Advances in digital instrumentation and fluorochrome chemistry have dramatically expanded the analytical capabilities of flow cytometry. Laboratories can now measure increasing numbers of parameters simultaneously. However, as analytical complexity has grown, so too has the need for structured standardization.
Collaborative initiatives such as the EuroFlow Consortium have played a key role by integrating standardized antibody panels, instrument calibration protocols, and computational analysis frameworks. Together, these efforts have improved reproducibility and diagnostic precision across laboratories.
High-Dimensional Cytometry and Panel Complexity
As cytometry moves toward higher-dimensional analysis, panel design has become increasingly complex. Laboratories must balance marker selection, fluorochrome compatibility, and instrument limitations while maintaining reproducibility.
To address this, computational tools such as FluoroFinder can support panel design by helping laboratories evaluate marker combinations and experimental constraints in a more systematic way. In turn, these approaches help translate complexity into more consistent and practical workflows.
AI and the Future of Clinical Cytometry
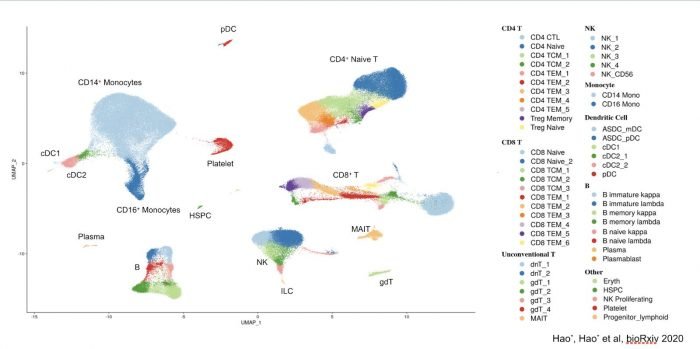
Figure: High-dimensional cytometry data visualized using clustering algorithms to identify immune cell populations.
Artificial intelligence is beginning to play a growing role in cytometric analysis. These tools can assist in identifying cell populations, recognizing patterns across large datasets, and supporting more consistent analytical workflows.
At the same time, in clinical environments these technologies remain assistive rather than autonomous. Their application must be carefully validated within established frameworks to ensure that analytical outputs remain accurate, reproducible, and clinically meaningful.
The Persistent Challenge of Variability
Despite advances in technology and analytics, variability remains a central challenge in clinical cytometry. It rarely appears as a dramatic failure. Instead, it manifests in subtle ways—minor shifts in reagent performance, gradual changes in instrument calibration, or small differences in gating strategies.
Over time, these small variations can influence interpretation, particularly in high-sensitivity assays. Maintaining consistency across changing conditions remains one of the most important responsibilities of clinical laboratories.
Looking Forward: Measuring Better, Not Just More
The evolution of clinical flow cytometry reflects a recurring pattern. Innovation expands what is possible, and standardization ensures that those capabilities translate into reliable clinical practice.
Ultimately, the central challenge remains unchanged: not simply to measure more, but to measure better. The goal is to produce results that clinicians can interpret with confidence and that patients can rely upon.
Behind every dataset is a clinical decision, and behind every decision is a patient. The continued success of clinical cytometry will depend on its ability to maintain trust in the measurements that underpin modern medical care.
About the Author
Paul E. Mead, PhD, SCYM(ASCP), is a Principal Scientist in the Department of Pathology at St. Jude Children’s Research Hospital and Technical Director of Clinical Flow Cytometry. His work focuses on clinical assay development, high-parameter spectral panel design, and translating cytometric measurement into improved diagnostic decision-making
References
- Brown M, Wittwer C. Flow cytometry: principles and clinical applications in hematology. Clin Chem. (2000) 46(8 Pt 2):1221-9.
- U.S.-Canadian Consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry. Bethesda, Maryland, November 16-17, 1995. Cytometry. (1997) 30(5):213-74.
- Stewart CC, Behm FG, Carey JL, Cornbleet J, Duque RE, Hudnall SD, Hurtubise PE, Loken M, Tubbs RR, Wormsley S. U.S.-Canadian Consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry: selection of antibody combinations. Cytometry. (1997) 30(5):231-5.
- Braylan RC, Atwater SK, Diamond L, Hassett JM, Johnson M, Kidd PG, Leith C, Nguyen D. U.S.-Canadian Consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry: data reporting. Cytometry. (1997) 30(5):245-8.
- Wood BL, Arroz M, Barnett D, DiGiuseppe J, Greig B, Kussick SJ, Oldaker T, Shenkin M, Stone E, Wallace P. 2006 Bethesda International Consensus recommendations on the immunophenotypic analysis of hematolymphoid neoplasia by flow cytometry: optimal reagents and reporting for the flow cytometric diagnosis of hematopoietic neoplasia. Cytometry B Clin Cytom. (2007) 72 Suppl 1:S14-22.
- Davis BH, Holden JT, Bene MC, Borowitz MJ, Braylan RC, Cornfield D, Gorczyca W, Lee R, Maiese R, Orfao A, Wells D, Wood BL, Stetler-Stevenson M. 2006 Bethesda International Consensus recommendations on the flow cytometric immunophenotypic analysis of hematolymphoid neoplasia: medical indications. Cytometry B Clin Cytom. (2007) 72 Suppl 1:S5-13.
- Bene MC, Castoldi G, Knapp W, Ludwig WD, Matutes E, Orfao A, van’t Veer MB. Proposals for the immunological classification of acute leukemias. European Group for the Immunological Characterization of Leukemias (EGIL). Leukemia. (1995) 9(10):1783-6.
- Craig FE, Foon KA. Flow cytometric immunophenotyping for hematologic neoplasms. Blood. (2008) 111(8):3941-67.
- Maecker HT, McCoy JP, Nussenblatt R. Standardizing immunophenotyping for the Human Immunology Project. Nat Rev Immunol. (2012) 12(3):191-200.
- Kalina T, Flores-Montero J, van der Velden VH, Martin-Ayuso M, Böttcher S, Ritgen M, Almeida J, Lhermitte L, Asnafi V, Mendonça A, de Tute R, Cullen M, Sedek L, Vidriales MB, Pérez JJ, te Marvelde JG, Mejstrikova E, Hrusak O, Szczepański T, van Dongen JJ, Orfao A; EuroFlow Consortium (EU-FP6, LSHB-CT-2006-018708). EuroFlow standardization of flow cytometer instrument settings and immunophenotyping protocols. Leukemia. (2012) 26(9):1986-2010.
- Keren DF, Carey JL, Braylan RC. Flow cytometry in clinical Diagnosis. 4th ed. (2007) Chicago: ASCP Press.
- Greig B. Flow Cytometry Certification. History and future plans. International Clinical Cytometry Society e-Newsletter. (2010) Vol.1 Spring.
- College of American Pathologists. Flow Cytometry Checklist. Northfield, IL: CAP Laboratory Accreditation Program.
- Clinical and Laboratory Standards Institute. Validation of flow cytometric Immunophenotyping Assays. CLSI guideline H62. Wayne, PA: CSLI (2021).
- Coustan-Smith E, Campana D. Immunologic minimal residual disease detection in acute lymphoblastic leukemia: a comparative approach to molecular testing. Best Pract Res Clin Haematol. (2010) (3):347-58.
- Theunissen P, Mejstrikova E, Sedek L, van der Sluijs-Gelling AJ, Gaipa G, Bartels M, Sobral da Costa E, Kotrová M, Novakova M, Sonneveld E, Buracchi C, Bonaccorso P, Oliveira E, Te Marvelde JG, Szczepanski T, Lhermitte L, Hrusak O, Lecrevisse Q, Grigore GE, Froňková E, Trka J, Brüggemann M, Orfao A, van Dongen JJ, van der Velden VH; EuroFlow Consortium. Standardized flow cytometry for highly sensitive MRD measurements in B-cell acute lymphoblastic leukemia. Blood. (2017) 129(3):347-357.
- Heuser M, Freeman SD, Ossenkoppele GJ, Buccisano F, Hourigan CS, Ngai LL, Tettero JM, Bachas C, Baer C, Béné MC, Bücklein V, Czyz A, Denys B, Dillon R, Feuring-Buske M, Guzman ML, Haferlach T, Han L, Herzig JK, Jorgensen JL, Kern W, Konopleva MY, Lacombe F, Libura M, Majchrzak A, Maurillo L, Ofran Y, Philippe J, Plesa A, Preudhomme C, Ravandi F, Roumier C, Subklewe M, Thol F, van de Loosdrecht AA, van der Reijden BA, Venditti A, Wierzbowska A, Valk PJM, Wood BL, Walter RB, Thiede C, Döhner K, Roboz GJ, Cloos J. 2021 Update on MRD in acute myeloid leukemia: a consensus document from the European LeukemiaNet MRD Working Party. Blood. (2021) 138(26):2753-2767.
- Borowitz MJ, Craig FE, Digiuseppe JA, Illingworth AJ, Rosse W, Sutherland DR, Wittwer CT, Richards SJ; Clinical Cytometry Society. Guidelines for the diagnosis and monitoring of paroxysmal nocturnal hemoglobinuria and related disorders by flow cytometry. Cytometry B Clin Cytom. (2010) 78(4):211-30.
- Sutherland DR, Keeney M, Illingworth A. Practical guidelines for the high-sensitivity detection and monitoring of paroxysmal nocturnal hemoglobinuria clones by flow cytometry. Cytometry B Clin Cytom. (2012) 82(4):195-208.
- Tangri S, Vall H, Kaplan D, Hoffman B, Purvis N, Porwit A, Hunsberger B, Shankey TV; ICSH/ICCS Working Group. Validation of cell-based fluorescence assays: practice guidelines from the ICSH and ICCS – part III – analytical issues. Cytometry B Clin Cytom. (2013) 84(5):291-308.
- Wood B, Jevremovic D, Béné MC, Yan M, Jacobs P, Litwin V; ICSH/ICCS Working Group. Validation of cell-based fluorescence assays: practice guidelines from the ICSH and ICCS – part V – assay performance criteria. Cytometry B Clin Cytom. (2013) 84(5):315-23.
- Centers for Medicare & Medicaid Services. (2024). Clinical Laboratory Improvement Amendments (CLIA): Laboratory quality standards (42 CFR Part 493). Balitmore, MD: CMS.
- Pui CH, Evans WE. A 50-year journey to cure childhood acute lymphoblastic leukemia. Semin Hematol. (2013) 50(3):185-96.
- Hunger SP, Mullighan CG. Acute Lymphoblastic Leukemia in Children. N Engl J Med. (2015) 373(16):1541-52.
- Giorgi JV, Detels R. T-cell subset alterations in HIV-infected homosexual men: NIAID Multicenter AIDS cohort study. Clin Immunol Immunopathol. (1989) 52(1):10-8.
- Mandy FF, Nicholson JK, McDougal JS; CDC. Guidelines for performing single-platform absolute CD4+ T-cell determinations with CD45 gating for persons infected with human immunodeficiency virus. Centers for Disease Control and Prevention. MMWR Recomm Rep. (2003) 52(RR-2):1-13.
- van Dongen JJ, Lhermitte L, Böttcher S, Almeida J, van der Velden VH, Flores-Montero J, Rawstron A, Asnafi V, Lécrevisse Q, Lucio P, Mejstrikova E, Szczepański T, Kalina T, de Tute R, Brüggemann M, Sedek L, Cullen M, Langerak AW, Mendonça A, Macintyre E, Martin-Ayuso M, Hrusak O, Vidriales MB, Orfao A; EuroFlow Consortium (EU-FP6, LSHB-CT-2006-018708). EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia. (2012) 26(9):1908-75.
- Nolan JP, Condello D. Spectral flow cytometry. Curr Protoc Cytom. (2013) 1:1.27.1-1.27.13.
- Ferrer-Font L, Pellefigues C, Mayer JU, Small SJ, Jaimes MC, Price KM. Panel Design and Optimization for High-Dimensional Immunophenotyping Assays Using Spectral Flow Cytometry. Curr Protoc Cytom. (2020) 92(1):e70.
- Liechti T, Weber LM, Ashhurst TM, Stanley N, Prlic M, Van Gassen S, Mair F. An updated guide for the perplexed: cytometry in the high-dimensional era. Nat Immunol. (2021) 22(10):1190-1197.
- Lee JA, Spidlen J, Boyce K, Cai J, Crosbie N, Dalphin M, Furlong J, Gasparetto M, Goldberg M, Goralczyk EM, Hyun B, Jansen K, Kollmann T, Kong M, Leif R, McWeeney S, Moloshok TD, Moore W, Nolan G, Nolan J, Nikolich-Zugich J, Parrish D, Purcell B, Qian Y, Selvaraj B, Smith C, Tchuvatkina O, Wertheimer A, Wilkinson P, Wilson C, Wood J, Zigon R; International Society for Advancement of Cytometry Data Standards Task Force; Scheuermann RH, Brinkman RR. MIFlowCyt: the minimum information about a Flow Cytometry Experiment. Cytometry A. (2008) 73(10):926-30.
- Aghaeepour N, Finak G; FlowCAP Consortium; DREAM Consortium; Hoos H, Mosmann TR, Brinkman R, Gottardo R, Scheuermann RH. Critical assessment of automated flow cytometry data analysis techniques. Nat Methods. (2013) 10(3):228-38.
- Saeys Y, Van Gassen S, Lambrecht BN. Computational flow cytometry: helping to make sense of high-dimensional immunology data. Nat Rev Immunol. (2016) 16(7):449-62.
- Ng DP, Simonson PD, Tarnok A, Lucas F, Kern W, Rolf N, Bogdanoski G, Green C, Brinkman RR, Czechowska K. Recommendations for using artificial intelligence in clinical flow cytometry. Cytometry B Clin Cytom. (2024) 106(4):228-238.