Written by Sarah Locknar
Flow cytometry controls serve several functions. They standardize and verify instrument performance, compensate for spectral spillover, verify specificity and sensitivity of staining, and help set gates for detection. Some controls are required for every experiment, while others are only required when changing batch numbers or setting up a new panel.
Flow Cytometry Controls for Clinical vs Research Panels
Clinical diagnostic panels are considered in-vitro diagnostics (IVD) by the FDA, and are highly regulated. The panels are fully validated by the manufacturer including sensitivity, specificity, precision and accuracy. Controls typically include daily instrument calibrations, single-stain compensation, positive and negative biological controls and limit of detection (sensitivity) determination. These panels also require strict Standard Operating Procedures (SOPs) including documentation (lot numbers, instrument settings, data storage protocols, etc.)
Research panels include those that are being developed for future clinical use and panels developed as part of a research project. FluoroFinder’s Panel Builder can be used to optimize the antibody/dye combinations specific for your flow cytometer instrument. Controls include instrument calibration, single‑stain compensation, positive and negative controls, unstained controls and reference samples to define gating and variability. Single-parameter assays with bright, well-separated populations typically only require unstained controls for background adjustment and instrument calibrations.
Essential Flow Cytometry Controls (used in both clinical and research panels):
1. Instrument Calibration Controls
Calibration with standardized fluorescent beads enables instrument monitoring without the added variables of staining reagents and protocols. Beads should be stored in the dark at low temperatures to ensure fluorophores do not bleach or degrade over time.
- Fluid flow, drift and optical alignment – Ensures beads (such as Alignflow™) are centered in the sheath liquid and aligned with the lasers and detectors in the system. Scatter detectors and/or fluorescence detectors can be used. Unstable flow rates and pressures can also lead to an increase in coefficient of variance (CV) during bead counting.
- PMT voltage adjustment – Adjusting the voltage helps maintain a constant mean fluorescence intensity (MFI) for bright particles and checks sensitivity of dim particles. An increase in voltage (or gain) may indicate scatter from a dirty flow cell or optical filter, a misaligned or failing laser, or failing optical filter. High voltages can increase the relative background in the measurement. MACSQuant® Calibration beads are used for automated voltage adjustment of Miltenyi Biotec’s flow cytometers.
- Detector linearity – Commercial beads typically come in at least three fluorescence intensities to verify that the detector’s response fits a linear regression at the optimal PMT voltage setting. Linearity falls off with very strong signals because of detector saturation. BD offers FACSDiva™ CS&T beads that are designed with the software to automate performance monitoring and traceability in their flow cytometers. These multi-labeled beads come in a mixture of three intensity levels for simultaneous sensitivity verification, linearity and standardization between instruments. Miltenyi Biotec offers MACSQuant® Advanced Calibration beads for voltage, detector linearity calibrations and compensation. The kit contains a mixture of multi-labeled (medium and bright), unlabeled and singly-labeled beads of different sizes for automatic calibration using their MACSQuantify™
- Image quality and corrections – For imaging flow cytometers, additional calibrations are performed to identify and remove bad pixels from the image, check focusing optics and acquire baselines for flat-field and other image corrections. BD CellView™ Calibration Beads include beads of several intensities to automatically calibrate the imaging system of the BD FACSDiscover™ A8 with BD CellView™ using the 488 nm laser. These beads are used instead of the CS&T beads.
- Standardize MFI readings between instruments or different days – The most rigorous method to standardize measurements between instruments is to use beads whose intensities are certified by NIST in Equivalent Reference Fluorophore (ERF) units. Calibrating MFI to ERF units enables the number of fluorophores per cell to be determined, independent of instrument. ERF values were originally determined using a set of four fluorophores, three excitation laser wavelengths and 29 different emission bandpass filters. In 2020 NIST added three more fluorophores and laser wavelengths with 50 bandpass filters.(DeRose, 2020) ERF values have been determined for beads in the 2-10 micron range.
ERF particle kits contain singly-labelled beads at several concentrations to generate accurate calibration curves against MFIs. They can also be used for detector linearity testing. ThermoFisher Scientific offers AccuCheck ERF Particles (watch the webinar), Bio-Techne offers an Ultra Rainbow Calibration ERF Particle Set and Spherotech offers Supra and Ultra Rainbow Quantitative Particles that are ERF characterized.
2. Single-color Compensation Controls
Compensation controls characterize the spillover values (SOVs) of fluorescence emission into unintended detection channels. SOVs depend on both the fluorophores and instruments being used. The specific lasers and filters installed in the system can vary in wavelength by a few nm due to manufacturing tolerances and age, which can lead to different SOVs. Read more about spectral spillover and compensation.
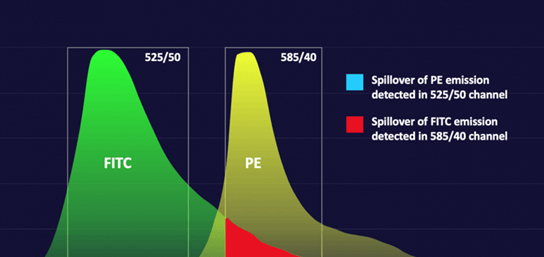
The best controls for compensation are sample cells labelled with a single fluorophore. This is especially true for imaging flow cytometry where the fluorescence intensity can vary significantly across the cell. If this isn’t possible because of sample scarcity, antibody capture beads may be used. It should be noted that this may take some trial-and-error because not all bead/fluorophore combinations are accurately compensated when compared to a cell-based method. (Bhowmick, 2023) A number of manufacturers sell antibody capture bead kits (containing positive and negative controls) including Thermofisher, Miltenyi Biotec, Abcam, Beckman Coulter, BD and Bio-techne.
For clinical systems (BD FACSLyric™), BD FC Bead kits which contain singly-labeled beads (and negative controls) are used to automatically calculate SOVs and create a compensation matrix. These kits are characterized at the factory with spectral overlap factors particular to that manufacturing lot.
3. Unstained and Autofluorescence Controls
Unstained controls are used to measure background levels for negative gating and autofluorescence compensation – required in traditional and imaging flow cytometers. Typically, the same cells to be analyzed are processed using the same protocol without the fluorescently tagged antibodies. This ensures there are no fluorescent contaminants in the reagents or other effects of the protocol on the fluorescence signal. Read more about autofluorescence in flow cytometry.
4. Biological Positive and Negative Controls
Biological controls are used to verify the reactivity of the antibody solution with desired antigens. Well characterized cells that are known to express the antigen at levels similar to the sample are used as positive controls. These can be known cell lines, transfected cells, or cells that have been induced to express the antigen. Negative controls include cell lines known not to express the antigen or cells that have had expression of antigens turned off with CRISPR or RNAi. Resources such as PAXdb and The Human Protein Atlas can help researchers determine anticipated concentrations of various antigens in their samples and to identify appropriate control cells.
Optional Flow Cytometry Controls
Reagent Controls
Similar to biological controls, reagent controls are used to verify that antibody and fluorophore solutions are working as expected and at the correct concentrations. An essential part of new panel development is titration of the antibody concentration so antigens are labeled with good signal-to-noise while non-specific binding is minimized. Any time a new batch of reagents is prepared, it is good practice to check the antibody titrations to ensure consistent results.
Isotype Controls
Isotype controls are a specific type of negative control that guards against Fc and other non-specific binding. Isotypes are antibodies from the same species, class (e.g. IgG1, IgG2a…) and fluorophore as the experimental antibody, but that do not intentionally bind target antigens. Target antigens are first blocked with non-fluorescent antibodies and then exposed to the fluorescent antibody isotype control. Any resulting fluorescent signal indicates non-specific binding that will need to be addressed with additional blocking protocols or gating strategies. Isotype controls are influenced by many variables, including specificity, concentration, degree of aggregation and fluorophore: antibody ratio, so they are of limited utility and are falling out of favor. (Maecker, 2006)
Fluorescence Minus One (FMO) Controls
Fluorescence minus one controls consist of a set of multiply labeled samples that each lack a single fluorophore-antibody conjugate. They are used to set a background level due to spectral overlap of multiple fluorophores. In a perfect world, compensation will mathematically remove all spectral overlap, but in practice, due to antigen concentration and binding variability between cells, a small amount of residual signal is often seen. This type of control is particularly relevant for gating weakly positive signals. They are generally not required in mature, validated panels if spillover and gating are stable and well documented. For researchers developing new panels or adding to existing ones, a full set of FMOs is recommended.
Viability Controls
Viability controls are fluorescent labels used to identify dead and dying cells. Dead cells have compromised cell membranes, so membrane-impermeable DNA-intercalating dyes are often used as labels. For sick and dying cells, fluorescently-conjugated Annexin V, which binds the apoptosis marker phosphatidylserine, and a DNA-intercalator are often used in combination. Protein binding dyes that bind primary amines can also be used as viability dyes. The concentration of primary amines is much lower on the cell surface than inside the cell, so fluorescence greatly increases when the cell membrane is compromised. These dyes are available in a number of wavelengths from many manufacturers including BioRad, ThermoFisher, Biotium and Miltenyi Biotec.
Summary
A robust strategy that combines the use of commercially available beads with cell-based controls helps monitor instrument and reagent performance while minimizing time preparing and analyzing control samples. ERF characterized beads have enabled standardization across instruments, and automated protocols simplify record keeping for clinical and cytometry core labs. Commercially available panels reduce the number of required controls overall, helping scientists make the most of their limited resources.
References
Bhowmick, L. R. (2023). doi:10.4049/immunohorizons.2300066
DeRose, T. E. (2020). doi:10.3390/ma13184111
Maecker, T. (2006). doi:10.1002/CYTO.A.20333
Supporting Your Research – Design and Optimize Your Multiplexed Fluorescence Experiments
The FluoroFinder Dye Directory and SpectraViewers are regularly assessed for accuracy and kept up to date. It is one of the only resources available to researchers without a bias towards the products of one company or another. Our goal is to show you ALL of your potential options, even from vendors you may be less familiar with but who may offer products that uniquely suit your application.