Written by Kelly Lundsten
It can be a challenge to navigate the reagent catalogs of vendors in the life sciences. New brand names are popping up all the time and it is difficult to know if they are truly emerging technologies or if they are simply the same chemistries being branded anew by different manufacturers. Here is a quick overview of the basic sorts of fluorescent chemistries you might find conjugated to antibodies along with their strengths and weaknesses to help.
What are Fluorophores?
All organic fluorescence, whether protein, polymer or multimer-based, must begin with the basic chemical building unit. Heterocyclic conjugated aromatic ring structures are like miniature conductive circuits that resonate energy through the shared pi cloud created by their double bonding.
The smallest structures, for example the metabolic cofactor NADH or the naturally occurring polyphenol molecule coumarin, are structures optimally excited by the shortest wavelengths of light, typically the ~355nm wavelength found in common cell analysis instrumentation like the UV laser or the principal peak of excitation from a metal halide arc lamp.
As a general rule, as the size of the fluorophore, specifically the surface of the conjugated ring structure, becomes larger, the optimal wavelength of absorption, excitation and emission also becomes longer, lower energy.
Organic Fluorophores
Attaching individual small organic fluorophores to a biomolecule has many advantages.
Due to their small size, they can often be engineered to be passively cell permeant. They are also less likely to cause steric interference in the binding of a biomolecule to its target, for example when labeling the terminal amine of a small peptide. They can also be easily engineered to either be more hydrophobic for lipid labeling or more hydrophilic for antibody conjugation or to shift wavelength or intensity under different environmental conditions like changes in pH or sensing analyte binding.
However, fundamentally, they will always have a limited potential brightness and, right now, we are limited to structures that are sensitive to wavelengths of excitation energy between 350-800nm. The largest simple organic fluorophore structures currently don’t exceed the 850nm range when it comes to their emission range. Near-infrared wavelengths are already very low energy. In addition, above ~800nm these fluorophores become increasingly insoluble.
Between these two factors, near infrared fluorophores cannot resonate energy efficiently enough or be conjugated to biomolecules at a high enough fluorophore to protein (F:P) ratio to be bright enough for applications in cell biology. Generally, there has not been any truly groundbreaking or novel technology in this chemical class of fluorophores in over 15 years. Although there are some instances where individual fluorophores have been made brighter, we have not managed to expand the useful spectral range of reagents.
This is also a reagent type where you may see multiple brand names unique to each vendor for essentially the same chemistries. Here is an example of fluorophores that all match the Alexa Fluor 594 spectral distribution. It is helpful when the vendor branding provides clues, for example many of these reagents contain 594 in the name, which refers to the excitation peak of these fluorophores. However, if you are curious whether the product you are interested in is similar to another, you can use a Spectra Viewer to overlay the spectra.
Products of the same basic chemical structures will overlay each other neatly, with little variance in their excitation or emission, like a fingerprint. However, there have been some advancements that have helped make some of these structures slightly brighter than others and that advantage is not something reflected in these spectral ribbons.
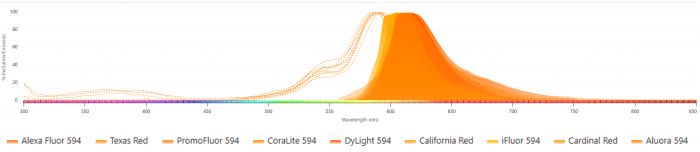
Figure 1. Overlay of excitation (dashed lines) and emission (solid lines) spectra for multiple fluorophores with Alexa Fluor 594–like profiles, demonstrating near-identical spectral distributions across different vendor brands.
How to Evaluate Fluorophore Brightness
There are different values you can try to collect from the manufacturer in order to assess potential differences in brightness between two spectrally similar products.
The first is the extinction coefficient of the fluorophore. The second is its quantum efficiency in an aqueous environment. Together, these two values would demonstrate any differences in the brightness of the single fluorophores but not the effective brightness of the biomolecule or antibody reagent.
For that, you would need to look at the certificate of analysis for information on the F:P (fluorophore to protein) ratio which can also be called its degree of labeling (DOL). But, let’s be honest, it would almost be easier to simply buy two similar products and empirically compare them side by side for performance.
Protein-Based Fluorophores
Protein-based fluorophores were likely some of the first fluorescent multimers produced by nature. Organisms like cyanobacteria/algae have a light-harvesting unit called phycobilins that capture light from the sun and transfer it to chlorophyll in a reaction center that facilitates photosynthesis.
In a single phycobiliprotein like the R-PE commonly used in flow cytometry, there can be 25-37 individual phycobilin fluorescent units. Another common protein-based fluorophore is green fluorescent protein and its genetic variants. Unlike the antenna system that cyanobacteria use to capture light, GFP serves a function in jellyfish that helps it adapt to the dark environment of its habitat.
Many fluorescent proteins made by marine organisms will naturally multimerize, assembling into structures that are excited by and emit longer wavelengths of light. For their use as protein reporters in life sciences research, it has been a challenge to engineer monomeric variants to prevent their multimerization which would impact protein localization and kinetics.
However, the massive expansion of spectral options in the last 15 years has had a dramatic impact on the ability to understand the complex cellular relationships in tissue, in vivo and real-time live-cell based research applications.
Protein-based fluorophores for antibody and biomolecule labeling are falling out of favor as synthetically created polymeric and multimeric fluorophores demonstrate significant advantages.
Polymer & Multimer Fluorophores
Fluorescent polymers and multimers are still only sold by the company that owns the brand or those with special licenses for resale. As they come off patent, I suspect you’ll see an explosion in the availability of generic forms of the polymers from other antibody vendors under different brand names.
BD Biosciences and Thermo Fisher Scientific are the only vendors who sell both the Brilliant UV and Violet fluorophores. In addition, BioLegend also helped developed and therefore sells the Brilliant Violet fluorophores. A few other companies offer polymer technology that is comparable to the Brilliant polymer family. Thermo Fisher sells a comparable product under the brand SuperBright and Beckman Coulter under the brand SuperNova. Bio-Rad offers their own unique chemistry called the StarBright fluorophore family that offers many advantages, especially as an alternative to Brilliant UV family and as a replacement for PE and APC tandems.
It is important to note that of all the polymer and multimer technologies, Bio-Rad is the only company to offer conjugation kits for their technology, which is such an important acknowledgement that not every researcher is a traditional immunologist who benefits from directly conjugated products. These conjugation kits are sold under the brand name TrailBlazerTM Tag. These polymer-based fluorophores all rely on a similar polyfluorine underlying chemistry.
A novel multimer technology from BD Biosciences has recently been released under the brand name Real, as in RealUV, RealViolet, RealBlue, RealYellow and RealRed, the color in the name indicating the laser of excitation. They offer many benefits in their reasonably small size, high solubility and lack of any unwanted non-specific binding issues as compared to polymers.
With each step in innovation that leads us towards reagents with less cross-bead excitation, high or tunable brightness and flexible emission peak choices, we are closer and closer to 50 color assays being accessible to the common flow cytometry user.
Researchers looking to multiplex any other immunoassay like fluorescence microscopy/confocal, cytometric bead arrays (CBA), spatial-omics and multiplex sandwich ELISAs should be taking note. These new generation of multimers have the potential to impact every fluorescent application.
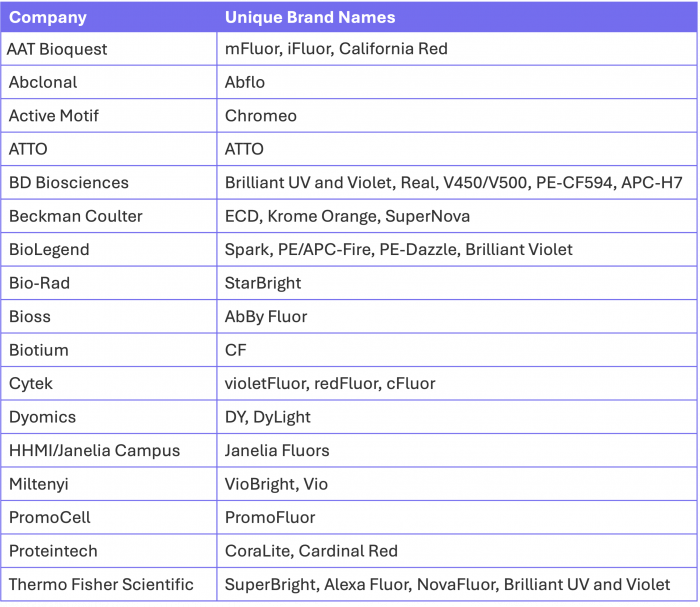
Fluorophore Brand Names by Vendor
Here is a quick guide, albeit likely not a totally comprehensive overview, of the most common unique brand names and their respective companies for your reference. This does not include generic fluorophores like Fluorescein, Texas Red, Cy5 and Cy5.5, to name a few, which are conjugates offered by most vendors. Some brands you may be more familiar with than others but it’s important to know that when it comes to simple organic chemistry, you have a wide array of options, especially when you are in search of antibodies not in the traditional immunology applications.
Table 1. Overview of common fluorophore brand names by vendor, highlighting the diversity of proprietary naming conventions for similar fluorescent dye chemistries.
Supporting Your Research – Design and Optimize Your Multiplexed Fluorescence Experiments
The FluoroFinder Dye Directory and SpectraViewers are regularly assessed for accuracy and kept up to date. It is one of the only resources available to researchers without a bias towards the products of one company or another. Our goal is to show you ALL of your potential options, even from vendors you may be less familiar with but who may offer products that uniquely suit your application.